Why Choose Us?
0% AI Guarantee
Human-written only.
24/7 Support
Anytime, anywhere.
Plagiarism Free
100% Original.
Expert Tutors
Masters & PhDs.
100% Confidential
Your privacy matters.
On-Time Delivery
Never miss a deadline.
Without referring to a data table, rank the following compounds by standard molar entropy Highest So Lowest S F2(g) Ne(g) HF(g)
Without referring to a data table, rank the following compounds by standard molar entropy Highest So Lowest S F2(g) Ne(g) HF(g)
Expert Solution
Part 1)
Compounds with smaller molar mass will have higher disorder and higher entropy
Here, order will be,
Ne(g) < HF(g) < F2(g)
Part 2)
A(s) + B(s) 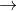 C(g)
C(g)
Here, solid reactants react to form the gaseous product. Therefore, this reaction will have a positive 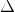 Srxn
Srxn
A(g) + B(g) 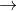 C(g)
C(g)
Here, moles of the reactants are higher than the moles of products. Therefore, this reaction will have a negative 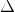 Srxn
Srxn
A(g) + B(g) 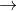 3C(g)
3C(g)
Here, moles of the reactants are lower than the moles of products. Therefore, this reaction will have a positive 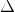 Srxn
Srxn
2A(g) + B(g) 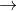 C(g)
C(g)
Here, moles of the reactants are higher than the moles of products. Therefore, this reaction will have a negative 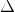 Srxn
Srxn
please see the attached file for complete solution
Archived Solution
You have full access to this solution. To save a copy with all formatting and attachments, use the button below.
For ready-to-submit work, please order a fresh solution below.







