Why Choose Us?
0% AI Guarantee
Human-written only.
24/7 Support
Anytime, anywhere.
Plagiarism Free
100% Original.
Expert Tutors
Masters & PhDs.
100% Confidential
Your privacy matters.
On-Time Delivery
Never miss a deadline.
The Ksp of PbBr2 is 6
The Ksp of PbBr2 is 6.60x10-6
What is the molar solubility of PbBr2 in pure water? Whats the molar solubility of PbBr2 in 0.500M KBr solution & whats the molar solubility of PbBr2 in a 0.500M Pb(NO3)2 solution?? Please explain
Expert Solution
Molar solubility is how many moles of a substance can be dissolved per liter of solution before the solution become saturated.
To determine molar solubility we need to look at the reaction of the substance dissociating into its component ions:
PbBr2(s) <--> Pb2+(aq) + 2Br-(aq)
Now, let's create an equilibrium expression for this reaction. An equilibrium expression expresses the concentrations of the products at equilibrium raised to the powers of their coefficients divided by the concentrations of the reactants at equilibrium also raised to the powers of their coefficients. BUT equilibrium expressions only include gaseous and aqeuous solutions (not solids or liquids) so in this case, our equilibrium expression would exclude PbBr2 giving us:
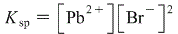
So let's determine their concentrations at equilibrium from an ICE table. When we create this table we're assuming we start of with zero of each:
PbBr2(s) <--> Pb2+(aq) + 2Br-(aq)
Initial / 0 0
Change / +s +2s
Equil. / s 2s
Where s represents the molar solubility of PbBr2. Plugging into our Ksp expression:
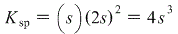
But we're given Ksp=6.60*10-6 so we get:
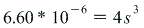
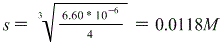
So that is our answer!
To find the molar solubility in a KBr solution, we do the exact same thing, except this time, when we create our ICE table we don't start with 0 Br-, we start with 0.500M Br- (KBr --> K+ + Br- so 0.500M KBr dissociates into 0.500M of EACH of it's component ions, which is why we start with 0.500M Br-)
So this time we have:
PbBr2(s) <--> Pb2+(aq) + 2Br-(aq)
Initial / 0 0.500M
Change / +s +2s
Equil. / s 0.500M+2s
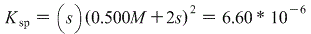
We can, however make this simpler by letting 0.500M+2s approximately equal just 0.500M, because the value of Ksp is soooo small (less than 10-4) that by adding that small of an amount won't affect our end result.
Therefore we get:
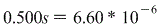
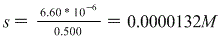
And again, s represents molar solubility so this is our answer!
For the molar solubility in a Pb(NO3)2 solution we do the same as above but this time we start with an initial concentration of Pb2+ ions. Pb(NO3)2 --> Pb2+ + 2NO3- so we start with 0.500M Pb2+
PbBr2(s) <--> Pb2+(aq) + 2Br-(aq)
Initial / 0.500M 0
Change / +s +2s
Equil. / 0.500M+s 2s
Giving us:
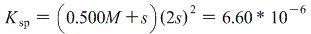
Again, we can let 0.500M+s approximately equal 0.500M for the same reason as above, giving us:
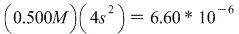
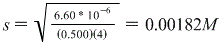
Archived Solution
You have full access to this solution. To save a copy with all formatting and attachments, use the button below.
For ready-to-submit work, please order a fresh solution below.







